No Analytes Found
Applications
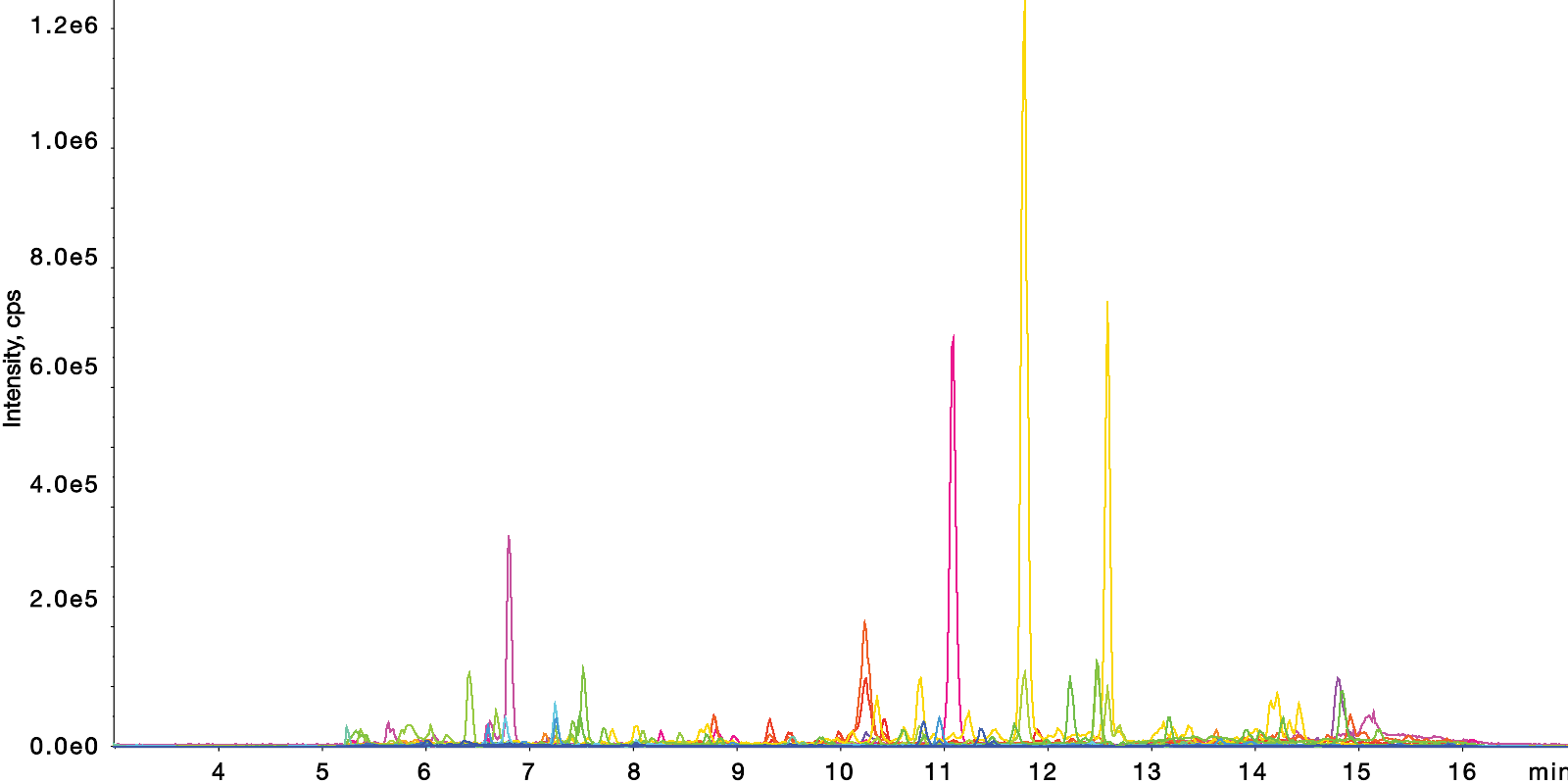
Signature Peptides of Rituximab on a bioZen Peptide XB-C18 Column Using bioZen MagBeads
25617
Separation Mode: Reversed Phase
Reversed Phase
PEPTIDE XB-C18
HPLC
Biopharmaceutical
Biopharmaceutical
Peptide
Biochemical Compound / Nutrient
Therapeutic / Clinical
Analytes
Details
LC Conditions (App ID: 25617)
Column: |
Brand Name: bioZen |
Part No: |
Phase Name: PEPTIDE XB-C18 |
System: Agilent Technologies SCIEX X500B Q-TOF |
Sample Note: *Rat |
Similar Applications
No Data