Biozen Oligo


Advanced Oligonucleotide Analysis
Biozen Oligo is an organo-silica, core-shell particle bonded with a C18 stationary phase. This column offers a unique combination of Core-Shell performance gains such as speed, sensitivity and resolution plus the high pH ruggedness necessary for oligonucleotide separations. Additionally, Biozen Oligo is packaged in BioTi™, a novel bio-inert hardware that serves to alleviate the sample loss and adsorption issues.
Increased Recovery and Reproducibility with NEW Biozen Oligo
- BioTi™ Hardware Reduces Sample Loss and Adsorption
- Robustness at High pH and Temperature
- Core-Shell Advantage for High Efficiency
Separation Mode Reversed Phase |
Particle Size 1.7, 2.6 |
Recommended Use Analytical characterization of oligonucleotides |
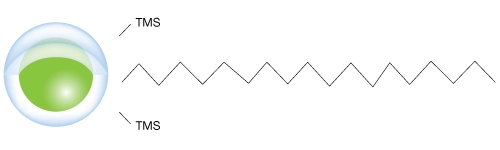
Patented Technology and Advanced Core-Shell Particle Chemistry
Biozen Oligo utilizes organo-silica core-shell technology to:
- Minimize band broadening associated with diffusion and oligonucleotide mass transfer kinetics
- Increase efficiency to produce narrower peaks, ideal for resolving closely eluting impurities
- Patented grafting process ensures high pH and temperature resistance from ethylene cross-linking.
BioTi versus Traditional Stainless Steel Hardware
LC-MS Analysis of Nusinersen
Oligos can chelate to trace heavy metals in stainless steel column hardware, leading to poor recovery, inconsistent chromatography and problematic carryover. The Biozen Oligo bio-inert hardware provides greater sensitivity as well as improved recovery, demonstrating this column’s optimal utility for oligonucleotide characterization and quantitation.
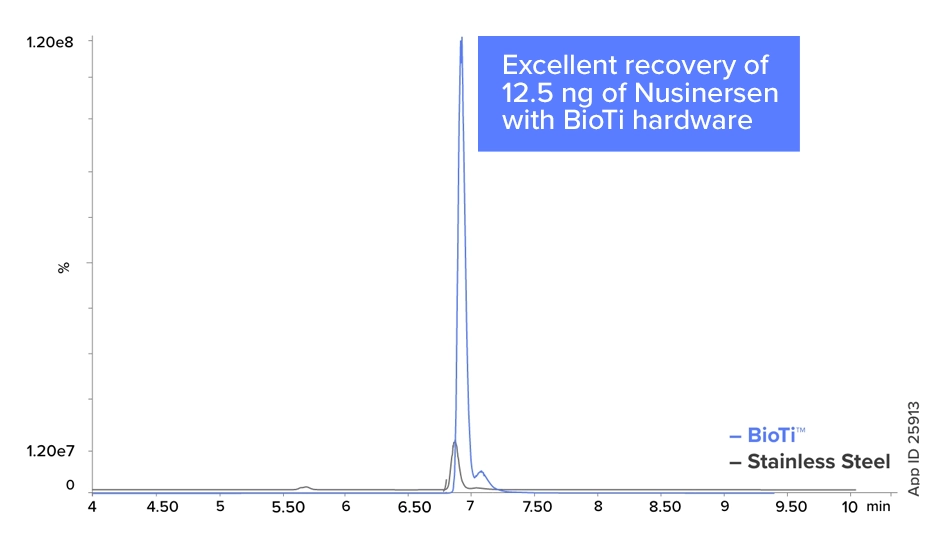
BioTi vs Stainless Steel Column Hardware
Biozen Oligo's bioinert hardware enhances recovery, sensitivity, and chromatographic reproducibility by mitigating non-specific adsorption. Stainless steel equivalents fails to prevent this resulting in poor recovery, peak tailing, and residual carryover.