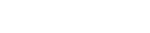| 1Lamivudine |
|---|
| 2Abacavir |
Applications
USP Abacavir and Lamivudine Tablets - Assay (Standard Solution)
28077
Separation Mode: Reversed Phase
Reversed Phase
C18
HPLC
Anti-HIV Agents,Reverse Transcriptase Inhibitors
Pharmaceutical
Pharmaceutical
Therapeutic / Clinical
Analytes
Details
LC Conditions (App ID: 28077)
Column: |
Brand Name: Luna Omega |
Part No: |
Phase Name: C18 |
System: Agilent Technologies 1200 Series Agilent Technologies 1200 Series |
Sample Note: 0.35 mg/mL of USP Abacavir Sulfate RS and 0.15 mg/mL of USP Lamivudine RS in 0.1N hydrochloric acid. Sonicate to dissolve prior to final dilution. Pass through a Nylon filter (Clarify Syringe filter PN: AF8-7707-12) |
Similar Applications
No Data